How does a limiting reagent affect a reaction

Daniel Johnson
Published Apr 02, 2026
The presence of a limiting reagent will reduce the amount of products a particular reaction can form. The reactant that acts as a limiting reagent will be consumed first by the reaction, in essence leaving the other reactant(s) in excess.
How does limiting and excess reactant affect a chemical reaction?
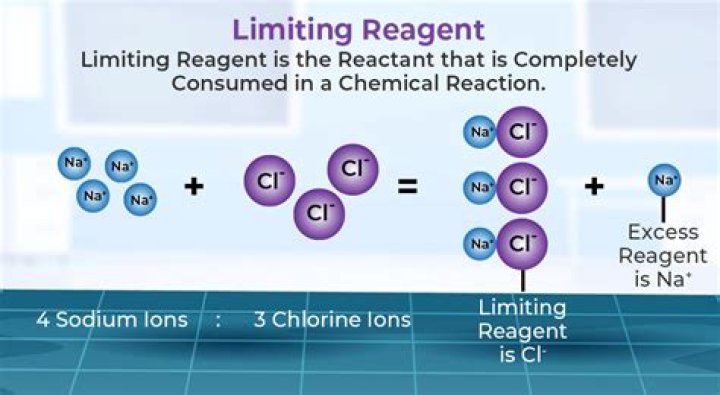
In a chemical reaction, reactants that are not used up when the reaction is finished are called excess reagents. The reagent that is completely used up or reacted is called the limiting reagent, because its quantity limits the amount of products formed.
How does a limiting reagent control the amount of product formed?
The limiting reagent (or limiting reactant or limiting agent) in a chemical reaction is a reactant that is totally consumed when the chemical reaction is completed. The amount of product formed is limited by this reagent, since the reaction cannot continue without it.
What does a limiting reagent do?
The limiting reagent is the reactant that is completely used up in a reaction, and thus determines when the reaction stops. From the reaction stoichiometry, the exact amount of reactant needed to react with another element can be calculated.What is the importance of knowing limiting reagent and excess reagent?
The limiting reagent in a chemical reaction is the reactant that will be consumed completely. Once there is no more of that reactant, the reaction cannot proceed. Therefor it limits the reaction from continuing. The excess reagent is the reactant that could keep reacting if the other had not been consumed.
What is limiting reagent explain with a suitable example?
The reactant which is entirely consumed in reaction is known as limiting reagent. In the reaction 2A+4B→3C+4D, when 5 moles of A react with 6 moles of B, then.
Why is excess reactant important?
A good way to ensure that one reactant fully reacts is to use an excess of the other reactant. This is financially efficient when one of the reactants is very cheap. The other reactant becomes a limiting factor and controls how much of each product is produced. …
Can the limiting reagent be a gas?
Since we have 2 moles of oxygen gas, some of it will remain unreacted when all of the hydrogen gas has been used. This means the hydrogen gas is the limiting reagent.When the limiting reactant in a chemical reaction is completely used up the?
The limiting reactant in a chemical equation is the reactant that is completely used up at the end of the reaction.
How does the maximum yield of products for a reaction depend on the limiting reactant?The amount of the limiting reagent determines the maximum amount of product that can be formed. The excess reactant is only partially consumed in the reaction. … It is always calculated using the limiting reactant. The limiting reactants determine the maximum yield they could be produced theoretically.
Article first time published onWhat is the limiting reagent in this reaction quizlet?
what is a limiting reagent? The reactant that determines the amount of the product that can be formed by a reaction.
What would be the benefit of having a limiting reactant when performing a lab experiment?
The reactant that becomes consumed is called the limiting reagent. When the limiting reagent is all consumed, no more product can be formed (reaction complete). The reactant limits the amount of product that can be formed.
Can there be a limiting reagent if only one reactant is present justify your answer?
No. Since the one reagent would be used up ‘first’ and would limit amount of product, then it is a limiting reagent. … One way to determine limiting reagent is to compare mole ratio of reactants used. This method is useful when there are only two reactants.
Why do many reactions slow down when the concentration of the reactants gets low?
Concentration: If there is more of a substance in a system, there is a greater chance that molecules will collide and speed up the rate of the reaction. If there is less of something, there will be fewer collisions and the reaction will probably happen at a slower speed.
Is the limiting reactant the one with less moles?
Explanation: The limiting reagent will be that with the lower quantity of moles . When we determine the limiting reagent, we first balance the chemical equation and convert all quantities of concern to moles. … That which is present in the lower number of moles is the limiting reactant.
Can you make the distinction between limiting and excess reagent?
The “limiting reagent” defines the maximum amount of product that can be produced from the reaction. The “excess reagent” is the one that will have some left in the solution after reacting completely (as much as possible) with the limiting reagent.
What happens when the limiting reactant runs out?
Whatever reactant runs out first is called the limiting reactant or limiting reagent. Thus B is the limiting reagent in the above scenerio. The reaction stops after the limiting reagent runs out. In the above case 1 mole of B reacts with 0.5 mol of A to produce 0.5 mole of AB2 with 0.5 mol of A unreacted.
Why is excess acid used in a reaction?
Using an excess of acid guarantees all the calcium carbonate in the eggshell reacts. If we don’t ensure this, our analysis will be inaccurate. The reaction may reach equilibrium and we want to use excess reactants to maximise the amount of product formed.
How do you know if something is a limiting reactant?
The reactant that is consumed first and limits the amount of product(s) that can be obtained is the limiting reactant. To identify the limiting reactant, calculate the number of moles of each reactant present and compare this ratio to the mole ratio of the reactants in the balanced chemical equation.
Which of the following is true about limiting reagent?
6. Which of the following is true about limiting reagents? Explanation: Limiting reagent is the reactant which reacts completely in a reaction. 7.
How is stoichiometry used in real life?
Stoichiometry continues to be useful in many walks of life—a farmer determining how much fertilizer to use, figuring out how fast you have to go to get somewhere in a specific period of time, or just to make conversions between systems like Celsius and Fahrenheit.
Which reactant hydrogen or oxygen is the limiting reactant in this case?
In this example, hydrogen is the limiting reagent and oxygen is the excess reagent. The amount of product formed is limited by the amount of hydrogen. In a chemical reaction, reactants that are not used up when the reaction is finished are called excess reagents.
How is a limiting reactant problem different from other stoichiometry problems What is your clue that it is a limiting reactant problem ?)?
A limiting reactant problem will most likely give the starting amounts of two or more reactants. The amount of products formed will be dependent on one of the reactants, but you won’t be told which reactant is going to determine the amount of products formed.
How can you identify the limiting reactant and excess reactant of a reaction quizlet?
The reactant that produced the lower amount is the limiting reactant. The reactant (or reactants) that produced the higher amount is the excess reactant.
What is excess reactant quizlet?
Excess Reactant. The substance that is not used up completely in a reaction, it is only partially consumed. Steps for finding the Limiting Reactant.
What is the significance of the limiting reagent in predicting the amount of the product obtain in a reaction?
Limiting reagent (also called limiting reactant) problems use stoichiometry to determine the theoretical yield for a chemical reaction. The limiting reactant will be completely consumed in the reaction and limits the amount of product you can make.
Why is limiting reactant essential in stoichiometric calculations?
Limiting reagent is essential in stoichiometric calculation because of the following reasons: It gives the information about the given chemical reaction and helps in a number of chemical calculations. It helps to calculate a number of products formed.
Why does a higher concentration increase the rate of reaction?
If the concentration of reactants is increased, there are more reactant particles moving together. There will be more collisions and so the reaction rate is increased. The higher the concentration of reactants, the faster the rate of a reaction will be.



