How do polyatomic ions work

John Castro
Published Mar 28, 2026
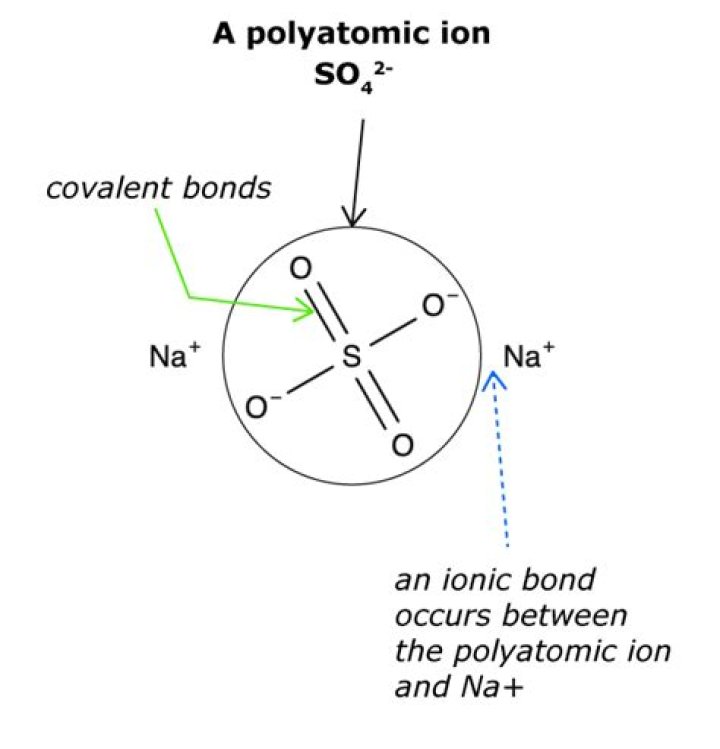
A polyatomic ion has two or more covalently bonded atoms that act as a single ion. The polyatomic ion forms ionic bonds with other ions and acts externally as a unit, just like monatomic ions.
How do polyatomic ion charges work?
In a polyatomic ion, the group of covalently bonded atoms carries a net charge because the total number of electrons in the molecule is not equal to the total number of protons in the molecule.
What polyatomic ion is io3?
Iodate is an iodine oxoanion and a monovalent inorganic anion.
Where do polyatomic ions get their charge?
Polyatomic ions get their charge by transferring electrons from another element. For example In NaNO3 the Na loses an electron and the NO3 gains the electron. Since then now have opposite ionic charges (the Na becomes Na+ and the NO3 becomes NO3-) the form an ionic bond.What is ClO polyatomic ion?
ClO- hypochlorite ion. S2O32– thiosulfate ion.
What element do most of the polyatomic ions have in the formula?
Most polyatomic ions contain oxygen.
How do you use ionic compounds with polyatomic ions?
Rule 1. The cation is written first in the name; the anion is written second in the name. Rule 2. When the formula unit contains two or more of the same polyatomic ion, that ion is written in parentheses with the subscript written outside the parentheses.
Why are polyatomic ions covalent?
Polyatomic ions are covalent compounds that have an overall charge. … According to valence electrons rule, the compound should have 9 valence electrons, 4 from hydrogen and 5 from nitrogen. But the compound contains eight electrons; it is missing one electron to give a charge of plus one.How do you read polyatomic ions?
Use the first letter (for N, C, S or P) or letters (for Cl) of the nouns in the sentence for the main atom in the polyatomic ion. For example, to write the formula and charge for nitrate, use N as nitrogen for “Nick.” Count the consonants in the word. This is the number of oxygen atoms in the polyatomic ion.
Is IO3 polar or nonpolar?Interestingly, among these six stoichiometrically identical compounds, α-(H3O)2Pt(IO3)6 is polar, whereas other compounds are nonpolar and centrosymmetric.
Article first time published onIs IO3 ionic or covalent?
IO3- has three bonds, so describing IO3- as a polar covalent bond is unclear. The I-O bonds are polar covalent bonds because of the electronegativity difference between the two atoms (which results in an unequal distribution of electrons).
What is io4 in chemistry?
Description. Periodate is a monovalent inorganic anion obtained by deprotonation of periodic acid. It is a monovalent inorganic anion and an iodine oxoanion. It is a conjugate base of a periodic acid. ChEBI.
Is clo1 an ion?
Hypochlorite is an ion composed of chlorine and oxygen with the chemical formula ClO−. Being unstable in the pure form, hypochlorite is most commonly used for bleaching, disinfectation, and water treatment purposes in its salt form, sodium hypochlorite.
What is common about most of the names of the polyatomic ions?
Summary. Polyatomic ions contain more than one type of atom in the ion. The majority of polyatomic ions are anions that are named ending in “ate” or “ite.”
Is Na+ a polyatomic ion?
For example, NaNO2 is named according to its cation, Na+ (sodium), and its polyatomic anion, … both polyatomic ions. • For example, NH4NO3 is ammonium nitrate.
Are polyatomic ions covalent or ionic?
Polyatomic ions are ions that are composed of two or more atoms that are linked by covalent bonds, but that still have a net deficiency or surplus of electrons, resulting in an overall charge on the group. A metal plus a polyatomic ion yields an ionic compound.
What keeps polyatomic ions together as a unit?
Polyatomic ions are ions which consist of more than one atom. For example, nitrate ion, NO3-, contains one nitrogen atom and three oxygen atoms. The atoms in a polyatomic ion are usually covalently bonded to one another, and therefore stay together as a single, charged unit.
How do you know if a polyatomic ion ends in ITE or ate?
H3O+hydroniumNH4+ammonium
Why do most polyatomic ions have oxygen?
They all have to gain electron to complete their octet. In almost every polyatomic ions we encounter oxygen which bears negative charge on it . Like in Hydrogen sulphide there is oxygen . So , polyatomic anions are generally formed.
How do you name polyatomic ions for dummies?
Ionic compounds involving polyatomic ions follow the same basic rule: Write the name of the metal first, and then simply add the name of the nonmetal (with the polyatomic anions, it is not necessary to add the -ide ending).
What type of bonding occurs in a polyatomic ion?
Covalent bonding is the type of bond that holds together the atoms within a polyatomic ion. It takes two electrons to make a covalent bond, one from each bonding atom.
What bonds do polyatomic ions form?
A polyatomic ion has two or more covalently bonded atoms that act as a single ion. The polyatomic ion forms ionic bonds with other ions and acts externally as a unit, just like monatomic ions.
Do polyatomic ions have intermolecular forces?
In contrast to intramolecular forces, such as the covalent bonds that hold atoms together in molecules and polyatomic ions, intermolecular forces hold molecules together in a liquid or solid. … Intermolecular forces determine bulk properties, such as the melting points of solids and the boiling points of liquids.
How many polyatomic ions are there?
The 9 polyatomic ions to know and write on your notecard:NameChargeFormulaPhosphate3-PO43-Hydronium1+H3O+Ammonium1+NH4+
How many atoms are there in polyatomic molecules?
Polyatomic molecules are electrically neutral groups of three or more atoms held together by covalent bonds.
What is the rule of zero charge?
You can use the positions of the elements on the periodic table to predict the ionic compounds they can form. When an ionic compound forms, the total charge on the atoms adds up to zero. This is known as the rule of zero charge.
Why does IO3 have pyramidal shape?
I has 3 single bonds with electron dense areas (O3) and a lone pair. If this lone pair was instead another oxygen molecule it would form a Tetrahedral shape. The lone pair makes the axial area on top unrepresented in shape making it Trigonal Pyramidal.
How many single bonds are in IO3?
IO3- lewis structure Oxygen atoms have made bonds with center iodine atom. From those bonds, there are two double bonds and one single bond in the IO3- lewis structure.
Is io3 soluble?
IdentifiersChemical formulaBa(IO3)2Molar mass487.13 g/molMelting point580 °C (1,076 °F; 853 K)(decomposes)Solubility product (Ksp)4.01 × 10−9
What is the hybridization of io3?
Further, during the combination of Iodine with the two other Iodine atoms, the central atom gains a negative charge whose value will be taken as 1. Therefore the hybridisation number is equal to 5. Now we can say that hybridisation is sp3d.
What Colour is io3?
NamesChemical formulaKIO3Molar mass214.001 g/molAppearancewhite crystalline powderOdorodorless



