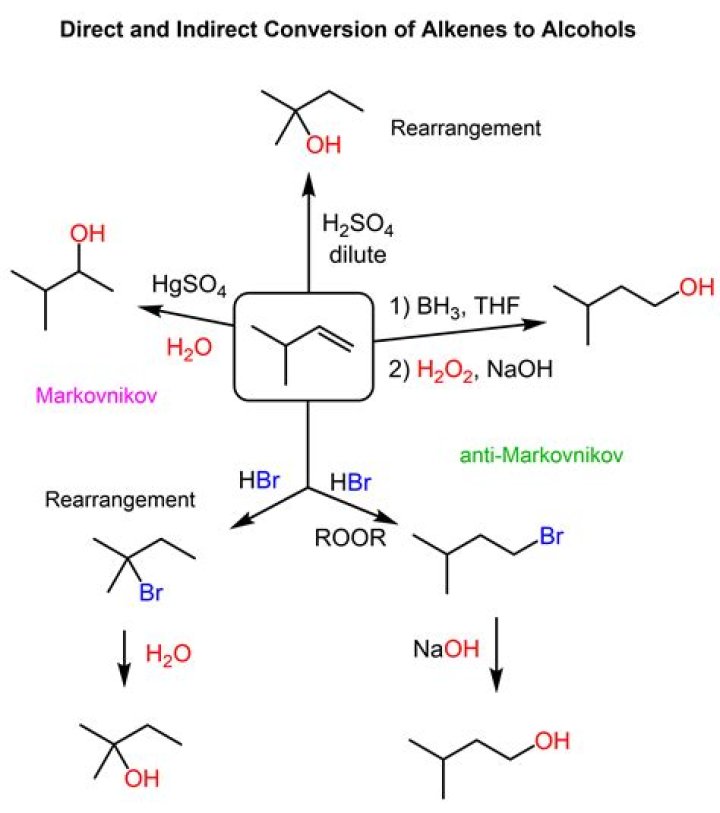
How do alkanes and alkenes react differently?

Daniel Johnson
Published Mar 14, 2026
How do alkanes and alkenes react differently?
Alkenes are relatively stable compounds, but are more reactive than alkanes because of the reactivity of the carbon–carbon π-bond. Most reactions of alkenes involve additions to this π bond, forming new single bonds.
What type of reaction is alkene to alkane?
One important alkene addition reaction is hydrogenation., where the alkene undergoes reduction to an alkane. In a hydrogenation reaction, two hydrogen atoms are added across the double bond of an alkene, resulting in a saturated alkane.
What are alkanes & alkenes explain?
Alkanes, alkenes and alkynes are simple hydrocarbon chains with no functional groups. Alkanes have only single bonds between carbon atoms and are called saturated hydrocarbons. Alkenes have at least one carbon-carbon double bond. Alkynes have one or more carbon-carbon triple bonds.
Which of the following reactions can both alkanes and alkenes generally undergo?
Alkanes undergo only substitution reactions but alkenes and alkynes undergo both substitution and addition reactions.
Which one is more reactive between alkane and alkene explain?
The number of hydrogen atoms in an alkene is double the number of carbon atoms, so they have the general formula. Alkenes are unsaturated, meaning they contain a double bond . This bond is why the alkenes are more reactive than the alkanes .
What is the main reaction of alkenes?
The most common type of reaction for alkene is the addition reaction to C=C double bond. In addition reaction, a small molecule is added to multiple bond and one π bond is converted to two σ bonds (unsaturation degree decreases) as a result of addition. Addition reaction is the opposite process to elimination.
What type of reactions are given by alkanes?
The result is that alkanes have very little reactivity and only undergo three major types of reactions, including the following:
- Combustion Reactions – burn them – destroying the entire molecule;
- Halogenation Reactions (substitution type) – react them with some of the halogens, breaking the carbon-hydrogen bonds;
Are alkanes electrophiles or nucleophiles?
Alkanes. Alkanes have no nucleophilic or electrophilic centers and are unreactive.
What are the reactions of alkanes?
Which type of reactions are given by alkanes?
Alkanes (the most basic of all organic compounds) undergo very few reactions. The two reactions of more importaces is combustion and halogenation, (i.e., substitution of a single hydrogen on the alkane for a single halogen) to form a haloalkane.
What is the most important reaction of alkanes?
The most important chemical reaction type involving alkanes is be combustion reactions which produces carbon dioxide and water (initially in water…
Why is alkyne more reactive than alkane and alkene?
Alkenes and alkynes are generally more reactive than alkanes due to the electron density available in their pi bonds. In particular, these molecules can participate in a variety of addition reactions and can be used in polymer formation.



