How did nitroglycerin crystallize

Christopher Anderson
Published Apr 01, 2026
At low temperatures, nitroglycerin exists in two crystal forms. It freezes to form a stable dipyramidal polymorph, which melts at 13.2°C. The compound is relatively stable in neutral and weakly acidic solutions, but degrades very rapidly in the presence of alkali.
What is crystalized nitroglycerin?
At low temperatures, nitroglycerin exists in two crystal forms. It freezes to form a stable dipyramidal polymorph, which melts at 13.2°C. The compound is relatively stable in neutral and weakly acidic solutions, but degrades very rapidly in the presence of alkali.
When did glycerin crystalize?
According to the game, in 1920, due to an accident involving the transport of liquid glycerin, a sample of glycerin crystallized. This was curious because glycerin had never been crystallized before, and afterwards glycerin everywhere started to crystallize spontaneously.
How did glycerin crystalize?
It has long been known that pure glycerin could be crystallized by special technics, involving super-cooling, followed by careful reheating or seeding with glycerin crystals. … organic liquids, such as liquid ammonia, or any of the various organic solvents for glycerin can be utilized.Can you solidify nitroglycerin?
Instability and desensitization Early in the history of this explosive it was discovered that liquid nitroglycerin can be “desensitized” by cooling to 5 to 10 °C (40 to 50 °F), at which temperature it freezes, contracting upon solidification.
How is a seed crystal made?
The large crystal can be grown by dipping the seed into a supersaturated solution, into molten material that is then cooled, or by growth on the seed face by passing vapor of the material to be grown over it.
How do you make nitroglycerin from glycerin?
Nitroglycerin is made by nitrating glycerin with a mixture of nitric and sulfuric acid. Glycerin is the backbone which bonds with fatty acids to make triglycerides, the main constituent of vegetable oils and animal fats. You can nitrate many molecules by mixing them with a mixture of nitric and sulfuric acid.
Can frozen nitroglycerin explode?
It is important to note that nitroglycerin freezes solid at 45-55 degrees Fahrenheit, depending on purity, and contracts upon freezing, unlike water which expands (water being an exception to the general rule). It is extremely insensitive to detonation in the frozen state as many stories in this series illustrate.Can nitroglycerin explode if frozen?
In its pure form, nitroglycerin is a contact explosive—that is, physical shock can cause it to explode. … Early in the history of this explosive it was discovered that liquid nitroglycerin can be “desensitized” by cooling to 5 to 10 °C (40 to 50 °F), at which temperature it freezes, contracting upon solidification.
Can you buy nitroglycerin liquid?Nitroglycerin is probably the most well-known liquid explosive. … Nitric acid, which can be bought at any Home Depot, and glycerin, another common household item, can also be combined to create an explosive.
Article first time published onHow can I make nitric acid at home?
Dissolve 80 grams (2.8 oz) of nitrate salt in 50 millilitres (1.7 fl oz) of water. Start by portioning out your nitrate salt into a small glass mixing container. Then, pour in all of the water at once. Swirl the mixture around inside the container to help it dissolve faster.
How do you get nitroglycerin?
- Enter Kyburz’s office in Hephaestus. The code is 0126.
- Disable the Trap Bolts and lower the plate glass. …
- Destroy the Turret and take the Nitroglycerin charge.
How do you make ammonium nitrate?
- Dissolve the sodium bisulfate in the mininum amount of water (about 300 ml).
- Dissolve your nitrate salt in the minimum amount of water (amount depends on the salt).
- Mix the two solutions.
- Next you want to neutralize the solution, which is quite acidic.
Can nitroglycerin be made from soap?
In the process of making soap, the fats and oils are hydrolysed (saponification), yielding glycerol (glycerine) and crude soap. … Nitric acid reacts with glycerine to make nitroglycerine but concentrated sulfuric acid is also needed to absorb the water produced.
What happens when glycerin is mixed with nitric acid?
The chemical combination of the glycerine with the acids results in the generation of intense heat, the glycerine as rapidly as it enters the chamber seizing the nitric acid, thereby combining to form nitro-glycerine, while, the sulphuric acid instantly absorbs the water which is released.
What neutralizes nitroglycerin?
In its purest form, nitroglycerin has a rather unique chemical structure that leads to it being extremely unstable and dangerous to handle. … 9 percent sodium chloride solution or five percent glucose solution, and this neutralizes the unstable and explosive effects of the nitroglycerin.
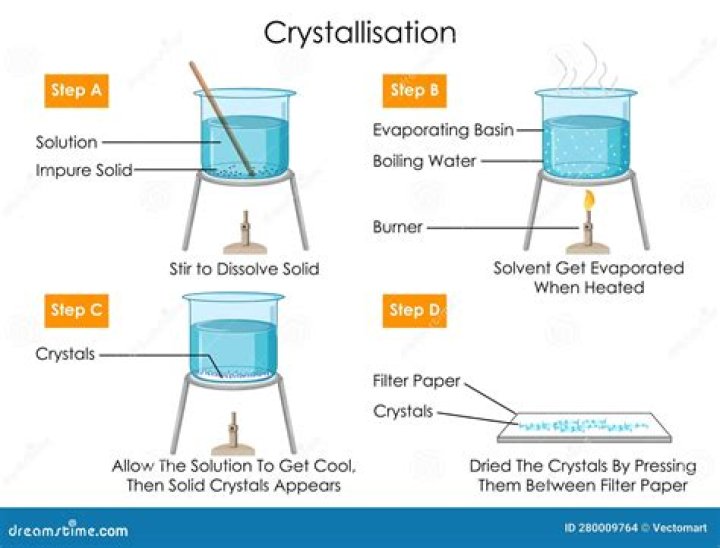
How do you induce crystallization?
Agitation during cooling may cause rapid crystallization, yielding less pure crystals. If no crystal formation is evident upon cooling, induce crystallization by gently scratching the inside walls of the flask with a glass rod or adding a small seed crystal of the compound being recrystallized.
How do you grow real crystals?
- In the beaker, stir 1/2 cup of magnesium sulfate with 1/2 cup of very hot tap water for at least one minute. …
- Add a couple drops of food coloring if you want your crystals to be colored.
- Put the beaker in the refrigerator.
- Check on it in a few hours to see a beaker full of crystals!
What are the three methods of crystallization?
- Evaporative crystallisation.
- Cooling crystallisation from solution or the melt.
- Precipitation.
What is c4 made of?
C-4 is made up of explosives, plastic binder, plasticizer and, usually, marker or taggant chemicals such as 2,3-dimethyl-2,3-dinitrobutane (DMDNB) to help detect the explosive and identify its source.
Who invented nitrocellulose?
The first practical smokeless powder made from nitrocellulose, for firearms and artillery ammunition, was invented by French chemist Paul Vieille in 1884. Jules Verne viewed the development of guncotton with optimism. He referred to the substance several times in his novels.
What is gelatinous dynamite?
In dynamite. Nobel also invented gelatinous dynamite, a mixture of nitrocellulose and nitroglycerin. Ammonium nitrate was later substituted for part of the nitroglycerin to give a safer and less expensive explosive called extra dynamite.
Is there nitroglycerin in TNT?
Dynamite is indeed an explosive with several components assembled together. But TNT (or 2,4,6,-trinitrotoluene, to use its chemical name) is not one of those components. Instead, the active explosive in dynamite is a chemical called nitroglycerin.
Does nitroglycerin smell sweet?
Pure nitroglycerin is a colourless, oily, somewhat toxic liquid having a sweet, burning taste. … It was first prepared in 1846 by the Italian chemist Ascanio Sobrero by adding glycerol to a mixture of concentrated nitric and sulfuric acids.
Is nitroglycerin a vasodilator?
Nitroglycerin is a vasodilator, a medicine that opens blood vessels to improve blood flow. It is used to treat angina symptoms, such as chest pain or pressure, that happens when there is not enough blood flowing to the heart.
What happens if you touch nitroglycerin?
* Contact can irritate the skin and eyes. * High levels can interfere with the ability of the blood to carry Oxygen causing headache, fatigue, dizziness, and a blue color to the skin and lips (methemoglobinemia). Higher levels can cause trouble breathing, collapse and even death.
Can nitroglycerin explode underwater?
Liquid nitroglycerine is colorless if pure. It is soluble in alcohols but insoluble in water. … This in turn, caused an explosion of the surrounding nitroglycerine. Alfred Nobel worked hard to improve nitroglycerine as an explosive that could be used in blasting rock and in mining.
What happens if you drink liquid nitroglycerin?
What if you had to swig a little nitroglycerine? You could get red in the face or faint. Nitroglycerine acts as a vasodilator to enlarge your blood vessels and lower your blood pressure. But screeners would have a hard time spotting the most likely symptoms—elevated heart rate and nausea.
How do I make aqua regia?
Fast Facts: Aqua Regia Aqua regia is a corrosive acid mixture made by combining nitric acid and hydrochloric acid. The usual ratio of acids is 3 parts hydrochloric acid to 1 part nitric acid. When mixing the acids, it is important to add the nitric acid to the hydrochloric acid and not the other way around.
Does vinegar and salt make hydrochloric acid?
When vinegar is mixed with salt, the acetic acid in the vinegar reacts with the sodium chloride or salt to produce sodium acetate and hydrochloric acid. The hydrochloric acid is a strong acid.
Is muriatic acid the same as nitric acid?
Muriatic acid is a totally different thing than nitric acid and substitutions are not possible. Nitric acid is an oxidizing acid, muriatic is not.