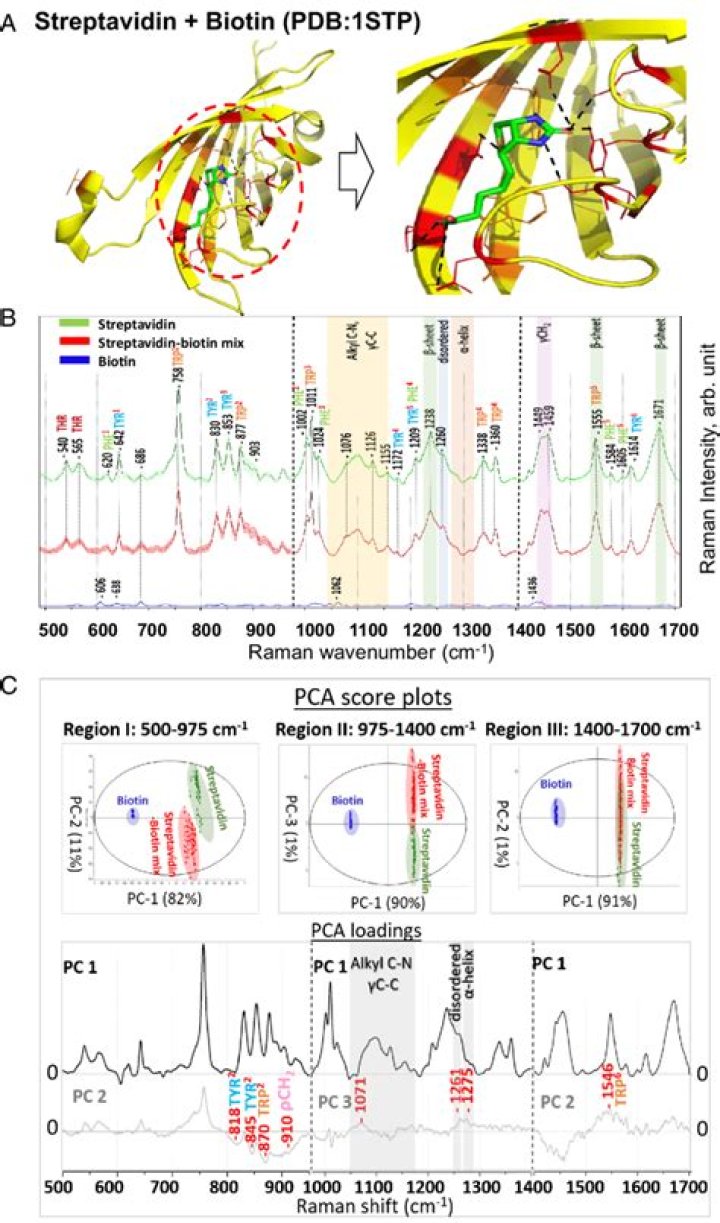
Does streptavidin bind biotin?

Daniel Johnson
Published Mar 01, 2026
Does streptavidin bind biotin?
Avidin, Streptavidin or NeutrAvidin Protein can bind up to four biotin molecules, which are normally conjugated to an enzyme, antibody or target protein to form an Avidin-biotin complex.
How many binding sites for biotin does streptavidin possess?
Once the streptavidin tetramer is bound to the surface of the MBs, there are two or three biotin-binding sites available for each streptavidin molecule on the surface of the bead.
What is streptavidin-biotin interaction?
The streptavidin-biotin system is a protein-ligand interaction present in nature that has been successfully used in a number of applications including detection of proteins, nucleic acids and lipids as well as protein purification.
How do you disrupt biotin streptavidin interaction?
The streptavidin-biotin bond can be broken efficiently only by harsh denaturing conditions. Hence, dissociation of biotin from streptavidin will denature both—the biotinylated protein and streptavidin, causing a leakage of the streptavidin monomer.
Where does streptavidin bind biotin?
β-barrel
The secondary structure of a streptavidin monomer is composed of eight antiparallel β-strands, which fold to give an antiparallel β-barrel tertiary structure. A biotin binding-site is located at one end of each β-barrel.
How many biotin molecules bind per monomer of streptavidin?
4 molecules
Since streptavidin is multivalent (binding 4 molecules of biotin per tetrameric protein molecule) it may be used in combination with biotinylated antibody and biotinylated reporter enzymes to obtain amplified signals.
How does streptavidin bind biotin?
Streptavidin is a tetramer and each subunit binds biotin with equal affinity. Multivalency is an advantage in applications like MHC tetramer staining, where avidity effects improve the ability of MHC molecules attached to streptavidin to detect specific T cells.
Does streptavidin bind DNA?
The protein of interest binds to the DNA, and then this complex binds (via the biotin moiety) to the tetrameric protein streptavidin. Since streptavidin is multivalent, it is able to serve as a bridge between the biotinylated DNA fragment and the biotin-containing resin.
What is streptavidin used for?
Streptavidin is widely used in Western blotting and immunoassays conjugated to some reporter molecule, such as horseradish peroxidase. Streptavidin has also been used in the developing field of Nanobiotechnology, the use of biological molecules such as proteins or lipids to create nanoscale devices/structures.
Is streptavidin biotin binding reversible?
A novel form of tetrameric streptavidin has been engineered to have reversible biotin binding capability. The biotin binding kinetic parameters (koff of 4.28×10−4 s−1 and Kd of 1.9×10−8 M) make this engineered mutein a superb affinity agent for the purification of biotinylated biomolecules.
How do you remove biotin from streptavidin?
The biotin can be cleaved from the labeled protein by the addition of DTT or TCEP. If you do not want to change anything in your system the most effective elution buffers from Streptavidin that have a chance of preserving protein function is . 5% Formic Acid in 30% acetonitrile.
What is the role of streptavidin in Elisa?
Biotinylated secondary antibody and streptavidin-HRP are commonly used in the ELISA to improve the limit of detection (LOD). Streptavidin is a tetrameric protein with a molecular weight of 60 kDa; it has a high binding affinity to biotin (in the low femtomolar range) and four biotin binding sites [36].