Does clay increase cation exchange capacity?

Christopher Anderson
Published Feb 28, 2026
Does clay increase cation exchange capacity?
Cation exchange capacity (CEC) is the total capacity of a soil to hold exchangeable cations. It influences the soil’s ability to hold onto essential nutrients and provides a buffer against soil acidification. Soils with a higher clay fraction tend to have a higher CEC.
Which cation has low adsorption capacity on clay?
Base-modified clay has lower adsorption capacities, while ZnCl2-modified clay had the least rate of adsorption with a surface area of 44.3 mm2 and porosity of 43.4 %.
Which clay mineral has the highest cation exchange capacity?
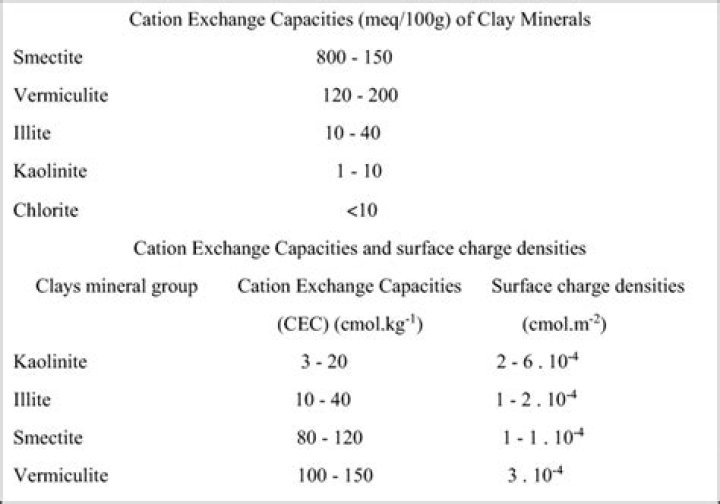
Clays such as kaolinite have a CEC of about 10 meq/100 g, while illite and smectite have CECs ranging from 25 to 100 meq/100 g. Organic matter has a very high CEC, ranging from 250 to 400 meq/100 g.
What is a good cation exchange capacity?
The cations are attached to the colloid by electrical attraction between the positive and negative charges. The five exchangeable cations are also shown in soil test results as percentages of CEC. The desirable ranges for them are: calcium 65–80% of CEC, magnesium 10–15%, potassium 1–5%, sodium 0–1% and aluminium 0%.
What is cation exchange capacity of clay?
Cation exchange capacity (CEC) is the amount of a cation that can be exchanged by another cation on the surface of a clay mineral. It is expressed in cmol(+)/kg, which is numerically equivalent to meq/100 g, where mol(+) represents moles of electrical charge.
Is high CEC good for plants?
Larger CEC values indicate that a soil has a greater capacity to hold cations. A high CEC soil requires a higher soil cation level, or soil test, to provide adequate crop nutrition. Low CEC soils hold fewer nutrients, and will likely be subject to leaching of mobile “anion” nutrients.
Which type of soil is best for adsorption of cations?
Soils are made up of sand, organic matter, silt and clay particles. Soils with high sand content have low holding capacity for cations compared to clayey and silty soils. Clay and silt particles have negatively charged sites which enable them adsorb and hold on to cations. Sandy soils have CEC between 1-5meq/100g.
Is anhydrous calcium chloride an adsorbent?
Both adsorbs and absorbs water molecule.
Is high CEC good or bad?
Larger CEC values indicate that a soil has a greater capacity to hold cations. These soils may benefit from split applications of several nutrients. The particular CEC of a soil is neither good nor bad, but knowing it is a valuable management tool.
What is a good CEC number for soil?
Optimum Percent Saturation Ranges
| Soil CEC | % K | % Mg |
|---|---|---|
| 0-5 | 4-6 | 10-20 |
| 6-10 | 3-5 | 8-20 |
| 11-15 | 3-4 | 8-20 |
| 16-20 | 2-4 | 8-20 |
What is a good CEC for soil?
Typical CEC Values in Soils
| Table 1. Cation exchange capacities at pH 7.0 of different soil types, textures and soil organic matter. | |
|---|---|
| Fine Sandy Loam | 5-10 |
| Loam | 5-15 |
| Clay Loam | 15-30 |
| Clay | >30 |
What is cation exchange capacity CEC of soil?
The total number of cations a soil can hold–or its total negative charge–is the soil’s cation exchange capacity. The higher the CEC, the higher the negative charge and the more cations that can be held. CEC is measured in millequivalents per 100 grams of soil (meq/100g).
What are the advantages of clay as an adsorbent?
The use of clays as adsorbent have advantages upon many other commercially available adsorbents in terms of low-cost, an abundant availability, high specific surface area, excellent adsorption properties, non-toxic nature, and large potential for ion exchange [50].
What cations are in clay soil?
Figure 1: Substitution of silica by aluminum in soil clay particles causes clays to have a negative charge. Because of this negative charge, the soil can hold on to positively charged cations such as calcium (Ca 2+), magnesium (Mg ) and potassium (K+).
Why are clay minerals used to adsorb metal?
The most of the clay minerals are negatively charged and very effective and extensively used to adsorb metal cations from the solution; due to their high cation exchange capacity, high surface area, and pore volume.
What is the cation exchange capacity of soil?
Cations are positively charged ions such as calcium (Ca2+), magnesium (Mg), and potassium (K +), sodium (Na) hydrogen (H+), aluminum (Al3+), iron (Fe2+), manganese (Mn2+), zinc (Zn 2+) and copper (Cu). The capacity of the soil to hold on to these cations called the cation exchange capacity (CEC). These cations are held by the negatively