Are protons and electrons equal?

Michael Henderson
Published Feb 28, 2026
Are protons and electrons equal?
An atom contains equal numbers of protons and electrons . Since protons and electrons have equal and opposite charges , this means that atoms are neutral overall.
How many electrons are in protons?
In other words, a neutral atom must have exactly one electron for every proton. If a neutral atom has 1 proton, it must have 1 electron. If a neutral atom has 2 protons, it must have 2 electrons.
How much of a proton is an electron?
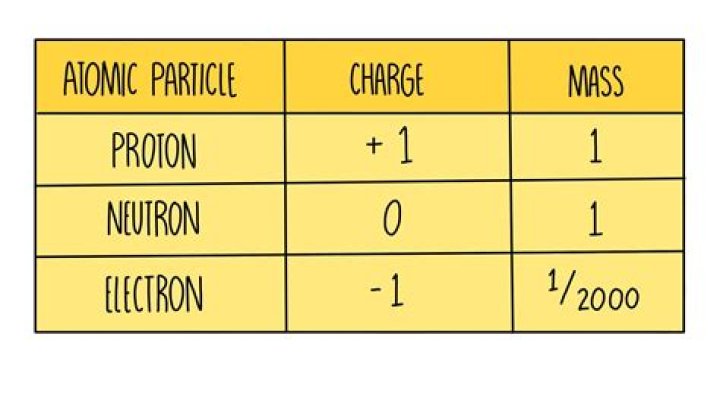
2.1 Electrons, Protons, Neutrons, and Atoms
| Elementary Particle | Charge | Mass |
|---|---|---|
| Proton | +1 | 1 |
| Neutron | 0 | 1 |
| Electron | −1 | ~0 |
How do you find the protons electrons and neutrons?
To calculate the numbers of subatomic particles in an atom, use its atomic number and mass number:
- number of protons = atomic number.
- number of electrons = atomic number.
- number of neutrons = mass number – atomic number.
Why is proton equal to electron?
Actually the proton and electron count of an atom are equal only when the atom is neutral in charge. The electrons are found in orbitals surrounding the nucleus. In order for the atom to remain electrically neutral the protons and electrons must balance each other.
What has 5 protons and 5 neutrons?
Boron
Atomic Number
| Name | Protons | Neutrons |
|---|---|---|
| Helium | 2 | 2 |
| Lithium | 3 | 4 |
| Beryllium | 4 | 5 |
| Boron | 5 | 6 |
Why are protons and electrons equal?
Where can you find the electron?
Where Are Electrons? Unlike protons and neutrons, which are located inside the nucleus at the center of the atom, electrons are found outside the nucleus.
Where do we find protons?
Protons: Positively charged subatomic particles located in the nucleus of an atom. Neutrons: Neutrally charged subatomic particles located in the nucleus of an atom. Electrons: Negatively charged subatomic particles located in orbitals surrounding the nucleus.
Why protons are positively charged?
Proton is made up of two up quarks(+4/3) and one down quark(-1/3) and due to this the net charge is +1 on protons and that is the reason protons are positively charged. While neutrons are made up of one Up quark(+2/3) and two down quarks(-2/3) due to this the net charge is 0(zero) on a neutron. Because of this n…
Can an atom have no protons?
Neutron matter is equivalent to a chemical element with atomic number 0, which is to say that it is equivalent to a species of atoms having no protons in their atomic nuclei.



